How many sigma bonds and pi bonds are there in c2carbon molecule Find the answer to this question along with unlimited Chemistry questions and prepare better for JEE 2020 exam. Want to see the full answer.
Is There A Picture Of How The Atomic Orbitals Overlapped In Co2 Quora
Does Sigma Bonding Occur In Co2.
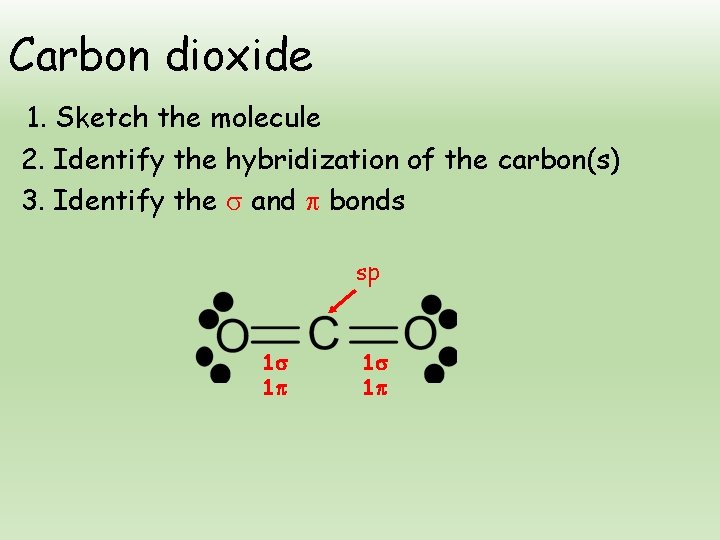
. There are two double bonds in carbon dioxide. Each double bond is comprised of one sigma bond and one π bond. Want to see the full answer.
S-S overlapping One s orbital from each participating atom experiences head-on overlapping along the internuclear axis in this type of overlapping. 3 sigma bonds and 2 pi bonds. A molecule has sp hybridization electron pair geometry.
The pi bond is the second bond of the double bonds between the carbon atoms and is shown as an elongated green lobe that extends both above and below the plane of the molecule. Predict hybridization of carbon atom in CH4 CH4 CH 7. Check out a sample QA here.
At low temperatures C_2 aggregates. A molecule has sp3d2 hybridization. Since CO2 has 2 double bonds it will have 2 pi bonds.
One sigma bond and one Pi bond combine to form a double bond. The CO2 bond has two double bonds so it has two pi bonds as well. Bonding in Carbon Dioxide From the Lewis structure we can see that the carbon in CO 2 must make 2 sigma bonds and it has no lone pairs.
Some of the properties of HCN are. It is important to note that a combination of sigma and pi bonds is always stronger than a single sigma bond. Generally double bonds consist of one sigma and one pi bond whereas a typical triple bond is made up of two π bonds and one σ bond.
The CO2 molecule has two pi bonds and two sigma bonds therefore. Carbon dioxide contains two double bonds. A faster way to determine how many pi bonds the molecule has is to know that a double bond is comprised of 1 sigma and 1 pi bond.
The sigma orbitals are quite the same as in ceCO2. Login Study Materials BYJUS Answer NCERT Solutions NCERT Solutions For Class 12 NCERT Solutions For Class 12 Physics NCERT Solutions For Class 12 Chemistry. Carbon dioxide is an important reagent used in industry is a cental aspect of our global carbon cycle and is the basis for climate change.
Pi bond is formed by natural overlap of p orbitals. Whereas it has a triple bond in C N and hence has two pi π bonds. Explain how the sigma and pi bonds are made in CO2.
- It can be synthesized by combining ammonia and methane. Explain how the sigma and pi bonds are made in CO2. According to VSEPR model predict the geometry of BrFs HO NH3 PCI XeF4 CIF.
The CO2 molecule has two pi bonds and two sigma bonds therefore. CO2 Sigma and Pi Bonds. How many σ sigma and π pi bonds are present in CO2.
The top and the lower half of the molecule does not have to be the same anymore hence the orbitals are more localised to one bond in particular. How many sigma and pi bonds in CO2. It is easier to determine how many pi bonds the sigma molecule has by knowing that it consists of one and one pi bonds.
Carbon dioxide molecules contain two sigma and two pi bonds so each double bond is made up of one sigma and one pi bond. How many sigma and pi bonds present in CH3COOH CHOHCO2H2SO4 CHCHCOOH 6. How many σ sigma and π pi bonds are present in CO 2.
It has two double bonds. This atom will be 2sp hybridized with remaining 2p x and 2p y atomic orbitals. Shapes of Organic Compounds Indicate the σ and pi bonds in the foll Question.
Indicate the σ and pi bonds in the following molecules. Pi Bonds are generally weaker than sigma bonds owing to the significantly lower degree of overlapping. But it does exist at very high temperatures and in the gaseous state.
In benzene the total sigma bonds are 12 and pi bonds are 3 hence the ratio is 41 In 13-butadiene there are c3 sigma bonds and 2 pi bonds hence the ration is 32 or we can say 151. It is easier to determine how many pi bonds the sigma molecule has by knowing that it consists of one and one pi bonds. O2NCH2CH2NO2 CH3COCH3 has CO HNO2 has OH Question.
Before one s orbital may overlap with. 1 sigma and 2 pi. TF Pi bonds are stronger than sigma bonds.
Seven sigma bonds and one pi bonds exist in CH2NCH3 How many double bonds are in CO2. What atomic or hybrid orbitals make up the sigma. Three sigma bonds are formed from each carbon atom for a total of six sigma bonds total in the molecule.
O2NCH2CH2NO2 CH3COCH3 has CO HNO2 has OH This problem has been solved. 2 C H 4 2 N H 3 3 O 2 Δ 1200 C P t 2 H C N 6 H 2 O. Count all the sigma and pi bonds in the following molecules.
2 sigma and 2 pi. Therefore CO2 has 2 pi bonds and 2 sigma bonds. Therefore the correct option is C.
So the HCN molecule has 2 sigma σ bonds and 2 pi π bonds. Check out a sample QA here. Each oxygen makes 1 sigma bond and also needs 2 orbitals for lone pairs of electrons.
C2H2 ethyne or acetylene HCCH has 2 sigma bonds between the carbon atoms and the terminal hydrogen atoms these are single bonds. Each double bond is comprised of one sigma bond and one π bond. A notably thing is that hydrogens take part in pi bonding Hyperconjugation.
Thus the number of sigma and pi bonds in C_2 molecule will be zero and two respectively. Determine the shape ideal bond angle s and the direction of any deviation from those angles for each of the following. C6H6 C6H12 CH2Cl2 CH2 C CH2 CH3NO2 HCONHCH3 Class 11 Chemistry Organic Chemistry - Some Basic Principles and Techniques Tetravalence of Carbon.
How many C-O pi bonds are in Methanol. This plane contains the six atoms and all of the sigma bonds. Usually most people think that C_2 molecule having 8 valence electrons does not exist.
In carbon dioxide there are 2 sigma and 2 pi bonds hence again the raio is 11.

0 Comments